sur SANOFI-AVENTIS (EPA:SAN)
FDA Approves Qfitlia for Hemophilia A and B Treatment
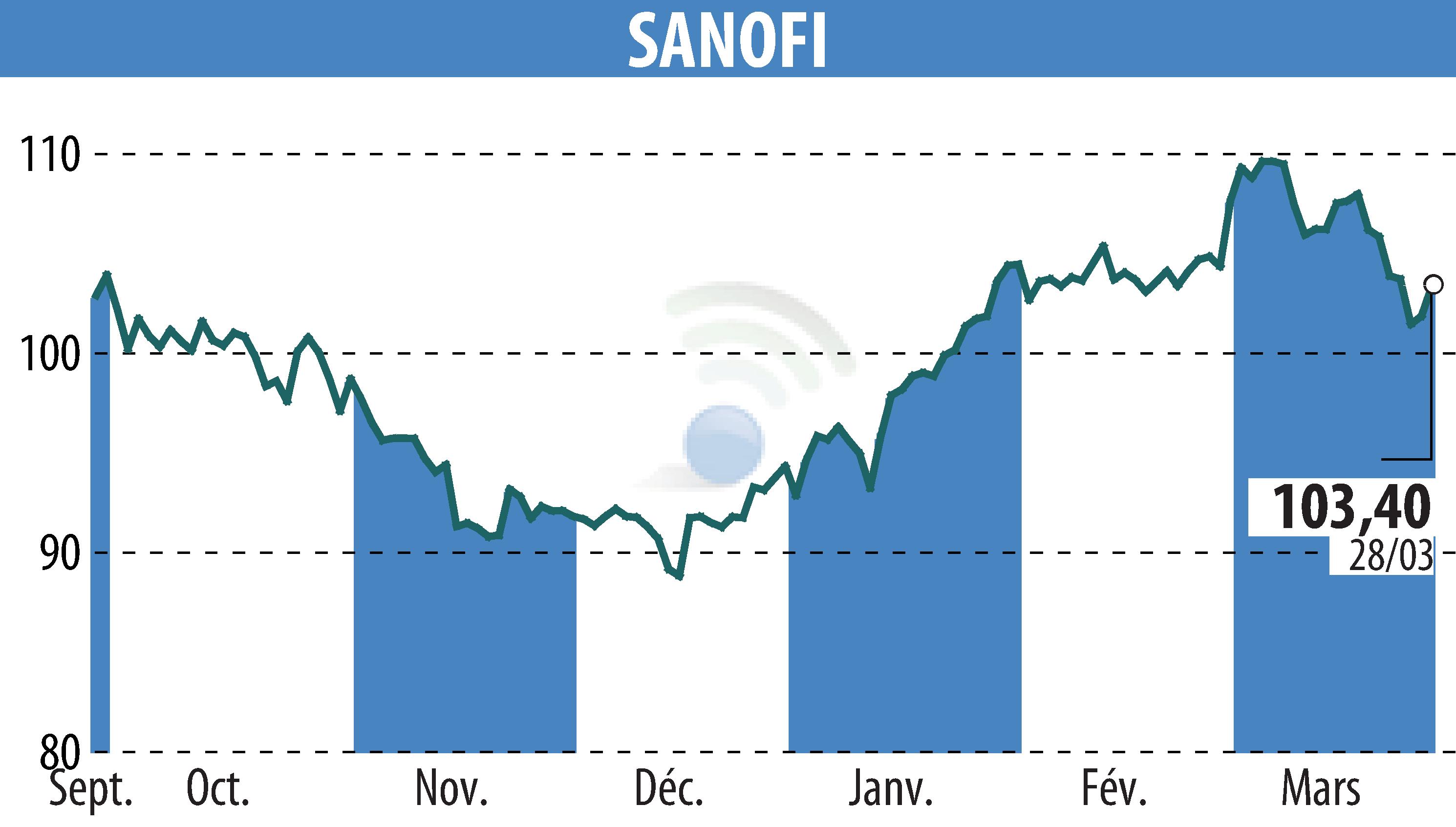
On March 28, 2025, the FDA approved Qfitlia (fitusiran), marking a significant milestone for hemophilia treatment in the US. Qfitlia is the first antithrombin-lowering therapy designed for hemophilia A or B patients, with or without inhibitors. The approval follows the ATLAS phase 3 studies, highlighting notable reductions in annualized bleeding rates (ABR) among different patient groups. Qfitlia's unique mechanism involves lowering antithrombin to enhance thrombin generation, thereby improving blood clotting.
Qfitlia requires as few as six injections annually, providing a less burdensome treatment schedule. Clinical trials revealed a significant 71% ABR reduction for patients without inhibitors compared to traditional therapies. However, potential adverse reactions include thrombotic events and infections.
The FDA has also approved a companion diagnostic assay to measure AT levels, enhancing treatment accuracy. Qfitlia is set to offer a comparable price to existing treatments, available with patient support services under the HemAssist program. Regulatory decisions in Brazil and China are anticipated in 2025.
R. E.
Copyright © 2025 FinanzWire, tous droits de reproduction et de représentation réservés.
Clause de non responsabilité : bien que puisées aux meilleures sources, les informations et analyses diffusées par FinanzWire sont fournies à titre indicatif et ne constituent en aucune manière une incitation à prendre position sur les marchés financiers.
Cliquez ici pour consulter le communiqué de presse ayant servi de base à la rédaction de cette brève
Voir toutes les actualités de SANOFI-AVENTIS
